The fundamental flaw in the lab leak conspiracy theory is the lack of a lab outbreak.More paper written by this Baric guy. This time it is written in 2016 and collaboration with some authors.
So these people studied a coronavirus from Chinese horseshoe bats, they mixed and match the genome of this virus with other known viruses such as the SARS1 virus to prove that it might have a pathogenic potential before nature can do it and even thanked Dr. Zhengli-Li Shi of the Wuhan Institute of Virology for access to bat CoV sequences and plasmid of WIV1-CoV spike protein.
Do we still have to believe that this covid virus is from natural origins?
Source:
A study led by researchers at the University of North Carolina at Chapel Hill found that a SARS-like virus known as WIV1-CoV, which is found in horseshoe bats, could bind to the same receptors as SARS-CoV and replicate in human cells without the need for adaptation. Thought to be a critical barrier, the results indicate that bat populations maintain SARS-like viruses poised to reemerge in humans.
The study, titled “,” was published in the latest issue of the Proceedings of the National Academy of Science (PNAS). The study’s primary investigator is Ralph Baric, PhD, professor of epidemiology at UNC’s Gillings School of Global Public Health.
The science paper:
Selected contents:
The discovery of SARS-like virus clusters that bridge the gap between the epidemic strains and related precursor CoV strain HKU3 virus provided the best evidence for emergence of SARS-CoV from Chinese horseshoe bats (). Comparing the receptor binding domain (RBD), SARS-CoV Urbani and WIV1 share homology at 11 of the 14 contact residues with human ACE2 (); importantly, the three amino acid changes represent relatively conservative substitution not predicted to ablate binding (). Therefore, exploring WIV1 strains allows examination of emergence, pathogenesis potential, and adaptation requirements. Using the SARS-CoV infectious clone as a template (), we designed and synthesized a full-length infectious clone of WIV1-CoV consisting of six plasmids that could be enzymatically cut, ligated together, and electroporated into cells to rescue replication competent progeny virions (). In addition to the full-length clone, we also produced WIV1-CoV chimeric virus that replaced the SARS spike with the WIV1 spike within the mouse-adapted backbone (WIV1-MA15, ). WIV1-MA15 incorporates the original binding and entry capabilities of WIV1-CoV, but maintains the backbone changes to mouse-adapted SARS-CoV. Importantly, WIV1-MA15 does not incorporate the Y436H mutation in spike that is required for SARS-MA15 pathogenesis (). Following electroporation into Vero cells, robust stock titers were recovered from both chimeric WIV1-MA15 and WIV1-CoV. To confirm growth kinetics and replication, Vero cells were infected with SARS-CoV Urbani, WIV1-MA15, and WIV1-CoV (); the results indicate similar replication kinetics and overall titers between the CoVs. However, Western blot analysis suggests potential differences in spike cleavage/processing of WIV1 and SARS-CoV spike proteins (); the ratio of full-length to cleaved spike varied between SARS spikes (Urbani, 1.21; MA15, 1.44) and WIV1 (full length, 0.61; WIV1-MA15, 0.25) signaling possible variation in host proteolytic processing (). Overall, the results indicate comparable viral replication, but possible biochemical differences in processing.
WIV1-CoV in Human ACE2 Expressing Mice.
Whereas studies in wild-type mice provide insight into pathogenesis potential, the absence of clinical disease in the epidemic strains of SARS-CoV suggests that the mouse model may not be adequate to access human disease potential. To test a model more relevant to humans, we generated a mouse that expresses human ACE2 receptor under control of HFH4, a lung ciliated epithelial cell promoter ().
A number of factors may contribute to reduced mouse pathogenesis observed following WIV1-CoV spike-mediated infection. In the context of both the SARS-CoV and MERS-CoV outbreaks, focus had been primarily directed to spike binding as the key component of emergence and pandemic potential. Supported by adaption at Y436H in mouse-adapted SARS spike (), improved binding to host receptor cannot be discounted as a crucial component in emergence. This fact is supported by improved replication of WIV1-CoV in mice expressing human ACE2 compared with control ( versus ). However, in vivo attenuation of WIV1-CoV relative to SARS-CoV Urbani despite efficient infection in primary human airway cultures suggests that additional factors contribute to epidemic emergence. One possibility is that adaptation outside of spike protein may lead to emergence via altered host–virus interactions.
Let's take a look at a map of the earliest cases. This can be found in Page 44 of the .
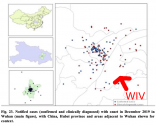
We clearly see that the outbreak starts in the western half of Wuhan, with the river acting as a geographical barrier. The Wuhan Institute of Virology is located distantly across the river, in the eastern half of the city.
If there was a lab leak, the initial outbreak would be on the left, centered around the Wuhan Institute of Virology. Instead it's on the right. The map is the opposite of what we would expect from a lab leak.